Not in this kind of market
https://twitter.com/KarunaTx/status/1769707862777823278
Exciting news! Today we completed our transaction with Bristol Myers Squibb. We look forward to continuing to transform the lives of patients with neurological and neuropsychiatric diseases as part of BMS. Follow our progress
Unit no 1 inspectio by USFDA
USFDA approves expanded labels for Esperion’s NEXLETOL and NEXLIZET, broadening treatment for heart attack prevention in primary & secondary prevention patients, regardless of statin use. Impacting ~70M patients in the US.
Credit Rating upgraded.
https://twitter.com/punitbansal14/status/1771832772597825696
https://twitter.com/rahuja671/status/1770119106261352896
https://twitter.com/rahuja671/status/1770119106261352896/photo/2
I’m sharing a GROUND report by Botlivala & Karani Securities Pvt. Ltd. on their visit to the Hyderbad Pharma plants. It also covers Neuland.
Pharmaceutical - Hyderabad Visit - Flash Note - 01 Apr 24.pdf (347.4 KB)
I hope you find it useful.
dr.vikas
Very detailed and indepth coverage on Neuland by BK
Some takeaways:
- CMS revenues will only go up from here
- Big supplies for Karuna will start after approval which is due in Sep’24
- Supplies to Esperion will go up
- Peptides things are slow but on track
- Capex as planned
- Aggressive hirings at R&D level
Key Policy Change that will accelerate the Indian CDMO Story Further!
The Biosecure Act would prohibit US companies receiving federal grant money from working with four Chinese biotech companies, including WuXi AppTec and its sister company WuXi Biologics, which produce active pharmaceutical ingredients (API) for hundreds of US and European drugmakers.
https://www.ropesgray.com/en/insights/alerts/2024/03/the-biosecure-act-a-review-of-the-bill-responses-and-possible-repercussions
https://twitter.com/soicfinance/status/1778484622348988647
FII 15.51% 16.03% 17.21% 17.21% 17.39% 17.68% 17.70% 18.14% 20.19% 21.57% 22.69% 24.43%
DIIs + 4.42% 5.65% 6.17% 6.38% 6.45% 6.57% 6.54% 7.10% 6.76% 6.99% 6.02% 6.54%
Increase in holding by institutions continues in Q4 also.
Neuland results: Drop at quarterly level, but excellent if looked at an annual level.
To be fair management has always guided to view results at an annual level or a longer time frame.
Stock is taking a beating now.
I feel the growth story is still intact . It’s a value migration theme from prime API to CMS and specialty. With biosecure act the opportunities will only improve. Balance sheet is also improving .
Disc : Invested and is the highest weight in portfolio.
Did anyone attend the conference call? Would be great if you could share key points/takeaways
Key takeaways from Q4FY24 Concall from own notes and screener:
Financial Performance:
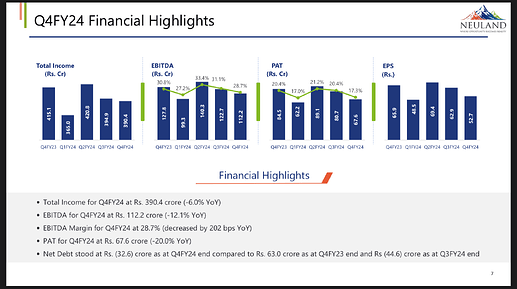
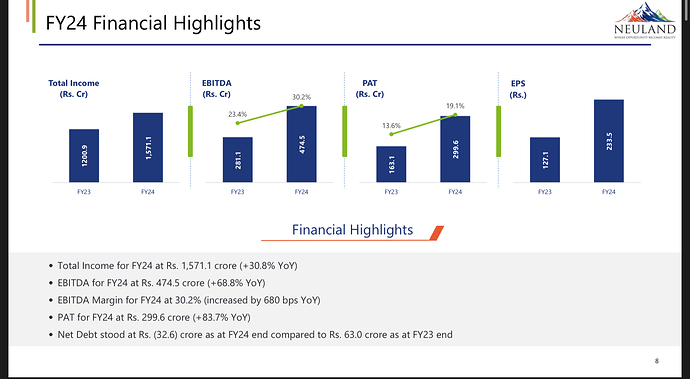
- Total income for FY24 was Rs. 1,571.1 crores, an increase of 30.8% from FY23.
- EBITDA for FY24 stood at Rs. 474.5 crores with a margin of 13.2%, an increase of 6.8% over FY23.
- Total income for Q4FY24 was Rs. 390.4 crores, with EBITDA at Rs. 112.2 crores and a margin of 28.7%.
- Gross margin was 58.8% in Q4FY24.
- Profit after tax was Rs. 67.6 crores in Q4FY24.
Operational Efficiency and Cost Optimization:
- Continuous work on optimizing costs and processes for long-term sustainability.
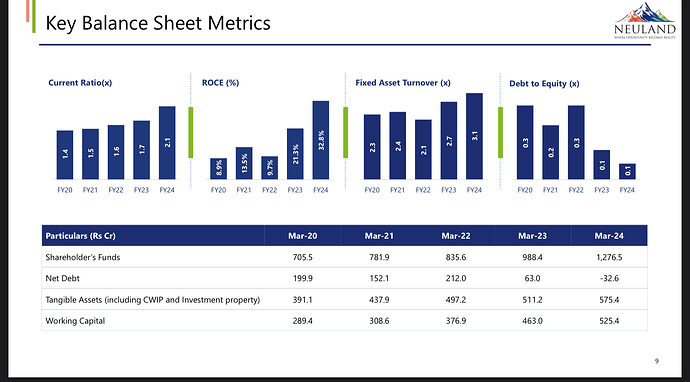
- Reduction of debt by Rs. 39.4 crores, leading to a net debt position of negative Rs. 32.6 crores.
- Working capital cycle reduced to 122 days at the end of March’24.
Capex and Investments:
- Invested Rs. 143.7 crores in capex during FY24.
- Continued investment in upgrading facilities to enhance business resilience.
Business Trends and Outlook:
- CMS business contributed close to 50% of revenues in FY24.
- Anticipating a surge in new projects in the next 12 months.
- Expecting a normalization of revenue growth and margin in FY25.
- Optimistic about 20% CAGR growth beyond FY25 based on current visibility of projects and portfolio products.
Customer and Industry Dynamics:
- Seeing a surge in RFI and RFP activities from customers.
- Customers considering Neuland differently due to track record and quality culture.
- Seeing increased interest from generic customers in the API space.
- Customers making strategic decisions on outsourcing based on various factors including geodiversity.
Biosecure Act and Industry Changes:
- Monitoring the developments around the Biosecure Act.
- Potential opportunities arising from the Act in terms of customer inquiries and RFPs.
- Preparedness to capitalize on opportunities and overcome challenges in the evolving regulatory landscape.
The concall was led by Saharsh and Sucheth (sons of the founder). After attending the last few concalls, I have noted that they are smart, realistic and down-to-earth management. They paint a very realistic picture of the future growth of the company and are very honest about what the company can and cannot do. They stayed away from speculation (especially around the Biosecure Act) and gave very practical answers based on their internal research and company’s expectations
Disc: Invested since much lower levels and plan to hold due to honesty and integrity of management.
Company’s management will be participating in the following group
analyst/ investor meetings:
Date Name Location
May 31, 2024 Trinity India B&K Securities Conference Mumbai
June 3, 2024 Nuvama India Conference 2024 London
Company’s management will be participating in the following group
analyst/ investor meetings:
Date Name Location
7-June-2024 Ambit Titans of Tomorrow Conference Mumbai
11-June-2024 Goldman Sachs Investor Day Hyderabad
Redefining Excellence: Neuland Unveils a Refreshed Brand Identity
Posted on June 7, 2024 by Saharsh Davuluri
In the pharmaceutical industry, change is an ever-present force. That’s why at Neuland, we recognize the need to not only keep pace with this evolution but to proactively lead it. Neuland is thrilled to announce the launch of its refreshed brand identity, marking a significant milestone in the company’s evolution. The new brand identity succinctly expresses our commitment to innovation and agility, ethical practices, strong partnerships, and sustainable growth embodying Neuland’s unwavering commitment to innovation, integrity, collaboration, and progress.

A brand for a healthier, brighter future
This new brand is more than a logo and color change. It marks a significant milestone for Neuland that reaches beyond our walls to directly impact our customers, our industry, and, ultimately, patients everywhere.
Here’s a glance into the logic behind this visual shift and how it expresses the heart of what we do at Neuland. Replacing our past logo (traditional type and historic mountain summits), our new look includes a refreshed, modern, bright mark that embodies the momentum of our industry and Neuland’s four primary business values our customers expect. They are:
- Agility and Innovation
Neuland’s new mark, while simplified, is layered in the story. The bright, green, modernized peak expresses our unwavering commitment to agility and innovation in the healthcare research space. This single peak represents both our history and our ability to scale new peaks in science as a leading provider of Active Pharmaceutical Ingredients (APIs) and Advanced Intermediates.
The mark’s upward trajectory signifies innovation and growth. There’s an intentional, forward motion designed into the mark that expresses Neuland’s passion to remain agile, innovate, and break barriers in complex chemistry for life-giving healthcare solutions.
- Ethical Practices
Our new mark also expresses a powerful simplicity around the concept of ethical practices. While we’ve always prioritized ethics, this commitment is now made visual with a more accessible, simplified brand identity.
With a focus on transparency, integrity, and compliance, Neuland reaffirms its dedication to upholding the highest ethical standards in every aspect of our operations. Simply put, we do what we say and stand by what we do in every step of the R&D journey. We see ethical practices as a responsibility and an ever-present value on which to build lasting trust with stakeholders, peers, and customers.
- Collaboration & Partnerships
Progress is impossible in science without trustworthy, collaborative partnerships. For that reason, it became imperative to us to design the interconnected, crossover strands as a central part of the Neuland logo. Our commitment to building strong partnerships with stakeholders, including healthcare professionals, research institutions, and other industry leaders—is now visually central and represented in everything we do. To us, powerful partnerships are critical to advancing pharmaceutical research, development, and patient care.
- Sustainable Growth
We’ve made a huge pivot in our brand with a whole new color palette. Why green? It’s simple. Green is at the root of who we are as scientists. It’s a color that expresses our commitment to sustainable growth, which, in turn, is the essence of life and why we do what we do.
It’s a color that clarifies Neuland’s goal of not only striving to operate a successful business but doing so in a way that is environmentally responsible and socially conscious. For that reason, we are actively pursuing initiatives that contribute to the well-being of communities and the planet, aligning our growth with the broader goals of sustainability.
Who We Are
If you had a trusted friend and partner in the API realm, ideally, that person would be trustworthy; they would be impactful, pursuing ideas that matter. They’d be tenacious and value what it means to be a devoted research and business partner.
In more than 40 years in this industry, that’s who we’ve worked to become at Neuland – a trusted partner for our stakeholders, our customers, and our team members. Our most recent rebrand has allowed us to identify and express the important character values of being trustworthy, impactful, and tenacious in everything we do.
Making a Difference: Together
Neuland’s mission is to be the leading, end-to-end resource for innovators in the pharmaceutical industry. We pride ourselves on delivering excellence to our customers and maintaining accountability and transparency in every interaction. We have been supporting Pharma and Biotech companies in custom small molecule API development and manufacturing for four decades. We specialize in facilitating and accelerating drug development and offering globally compliant manufacturing facilities.
Are you ready to change the world? Our experienced team can help you navigate the complex components of pharma R&D, tech transfer, IP, project management, quality control management, and regulatory compliance. To partner with us and learn more about The Neuland Way, connect with our team today.
.
Comments are closed.
“Sharing the latest tweet by Sajal Kapoor sir on Neuland.”
https://x.com/unseenvalue/status/1805553798577795407
I hope you find it useful.