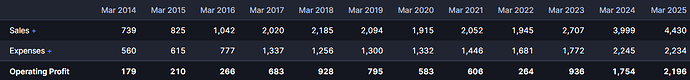Thanks for sharing your thoughts @madhavojha. Here is my analysis:
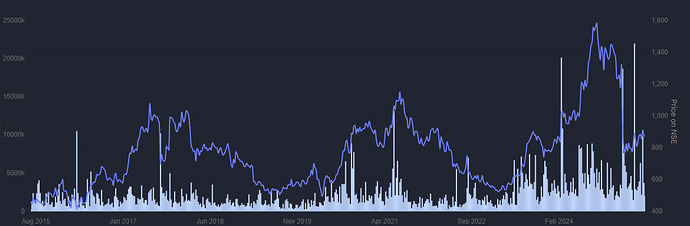
Revlimid sales will decline as patent expires at end of CY25 and so sales and PAT will decline in FYe26. Management has guided for PAT of ~1250 Cr in FYe26 on sales of ~3550 Cr. Revlimid sale price per unit and margins will decline much further for FYe27 as competition sets in and price erosion happens.
But there are five factors might help alleviate this decline:
(i) while margins decline, market share of Revlimid innovator Bristol Myers Squibb should go down as quotas are no longer applicable. This can lead to increased volume for generic players like NATCOPHARMA, thereby offsetting to some extent the decline in margins.
(ii) successful launch(es) of the many first to file approvals (sole and joint) that the company has, including Olaparib, might help offset this decline
(iii) successful launch of Semaglutide in India
(iv) Their agrochemicals business turns profitable,
(v) with cash reserves ~3500 Cr a good acquisition should be EPS accretive. The average return on equity (RoE) of Indian pharma companies is ~13% while FD rates are say 6%. In that case an average acquisition might boost PAT by 7% of 3500 Cr = 245 Cr which corresponds to EPS of ~13.
In any case there is likely to be very significant volatility/lumpiness in sales/profits as some drug in their current portfolio wanes and then it takes time for a new launch to gain traction. Over a longer period also there will be significant volatility/lumpiness in sales and/or profits as existing products face price erosion and new launches take time to ramp up. Overall, to me the three scenarios are:
(a) In the bear case scenario, none of the above 5 factors fire and EPS drops to 70 in FYe26 and 50 for FYe27.
(b) Baseline is EPS of 75 in FYe26 and further decline is arrested due to one/two upside factors playing out leading to EPS of 75 in FYe27.
(c) Bull case scenario would be that three/four of the above 5 factors play out favourably leading to EPS of 80 in FYe26 and rebounding to 105 in FYe27 (i.e. to FYe25 levels).
It seems to me that the management has a good reputation for integrity and that gives me much more comfort than the above scenario-based projections. Would be happy to hear other people’s thoughts and/or observations.
Disclosure: invested and possibly biased.