I did not see this disclosure FDA Confirms Recalls of High Blood Pressure, Heart Drugs Over Carcinogens | The Epoch Times
Further to my last post on inlicensing above today Glenmark has announced inlicensing of hengrui’s shr-a1811
It is currently in phase ii/iii and has high efficacy and safety data.
Application in breast cancer , lung cancer, gastri, esophageal , colorectal etc
breast cancer market alone for her2 therapies is approx 13 bn usd.
One more inlicensing deal done for oncology. The drug is a sucess in china with sales of 40 bn RMB approx 1200cr pa. They are going to be big in oncology in ROW and India by the number of novel drugs inlicensed and developed inhouse (isb 2001) in their portfolio.
There is a possibility that isb 830 which they had outlicensed to astria may be bought back, as astria has a change in mgmt and new mgmt is focused on other pipeline drugs of astria. Glenmark is aware of the possibilities of isb 830 and by some statements might bid for the asset. Check grok or other AI tool for possible commercial value of isb 830.
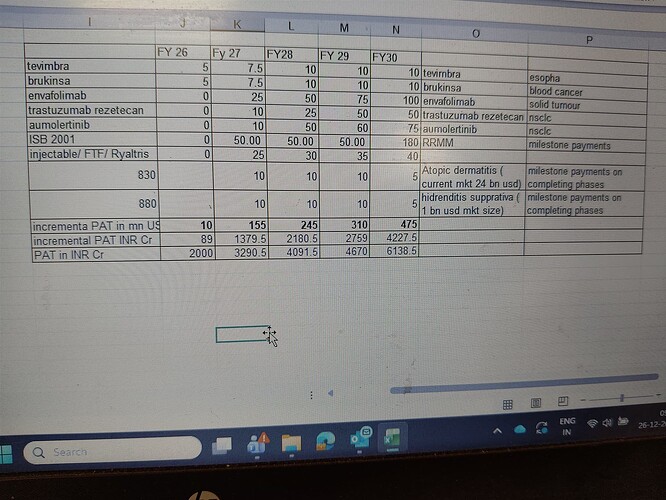
Estimated increase in PAT due to pipeline drugs… timeline of milestone payment and amount is rough conservative estimate basis public information
Like isb 2001milestone payment for isb 2001 will be 1.2 bn usd in total, so for phase 1,2,3 it should be roughly 200 mn usd each and balance on marketing Authorization but I have taken only 50 mn each
Further detailing the injectables/ para iv/ biosimilars filing we have pipeline of gvoke, iron deficiency, etar ncept, trastazumab, bevacizumab etc mainly for usa and ROw.
Europe has been doing well but the pipeline looks weak as of now. India, Row & usa pipeline has something or the other.