I am not speculating. Just trying to understand the reason for 20% fall from highs despite having good results. Valuation could be one of the reason that Avanti is falling but others are not. Avanti has increased 5-6 times in last one year but others have hardly increased 2 times.
Avanti has a fantastic capital structure. I would assign it a base PE of 23. If it can maintain its growth rate then a fair PE in my view is in the 35 range.
However, investors in Avanti are and will forever be plagued by visibility issues due to the inherently unstable nature of the industry it operates in. It will be difficult even for the most seasoned investors to have a view that extends for more than 3 years.
If Avanti forms a major part of your portfolio then I would advise that one forms a group of people that consistently does ground checks across the value chain Avanti operates in to have an edge on the visibility part. Once a system of ground checks is established I think it will help insulate everyone from the adverse newsflow. On the flip side one can also sense any threats in advance. Either ways I think given the interest in the company it would be well worth the effort.
Best
Bheeshma
Disc - not invested due to the aforementioned reasons.
Does anyone know if concall is scheduled / date of concall?
Regarding the EU officials visit, I have few questions on the bearish scenario:
- EU officials increased the samples for testing from 10 to 50% and now as per one of the above article they may increase to 100%. Do we have the rejection rate from Avanti ?
- In worst case, what if EU bans imports from India ? How does this gonna affect the SECTOR and Avanti?
This is quite unlike Avanti…
But good that management is now getting investor savvy
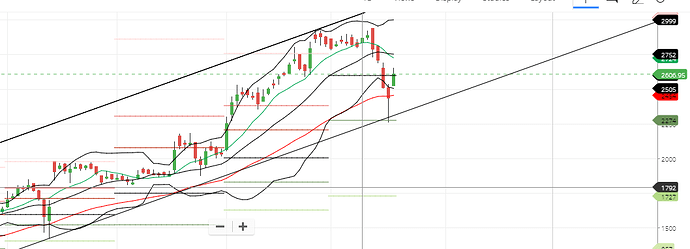
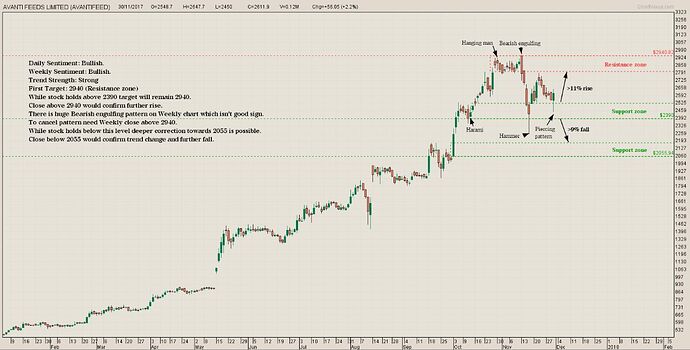
Avanti just bounced back from its channel support:
It would be interesting to see if it reaches its channel resistance at 3150.
Added at 2300 levels on 11/16.
Conf Call details and presentation for Q2 results.
http://www.bseindia.com/xml-data/corpfiling/AttachLive/4a83b7ce-7f44-461e-b61b-2b1fdbf7b4df.pdf
Management confidence of $ 1 Bn by 2022 augurs well for the growth of the company. That means in today exchange rate, the turnover is expected to be around Rs 6500 Cr, implying 150% increase in turnover in 5 years.
Avanti Feeds won the Forbes India Leadership Award 2017 for Value Creator category
Good read - http://www.forbesindia.com/article/leadership-awards-2017/avanti-feeds-harvesting-high-values/48695/1
Key highlights (non-exhaustive) -
- Avanti Feeds is one of the fastest growing mid-cap companies in the Indian market and investors feel there is still a lot of steam left.
- In 1993, the company had a market share of 7 percent, which has gone up to 45 percent today.
- Investors prefer companies that have a global market and one that grows fast. According to a 2013 World Bank research paper, aquaculture is the fastest growing form of food production in the world
- The global market has huge potential for Indian aquaculture and that is the reason why many shrimp farming capacities are being added across the nation, especially in new places like West Bengal and Haryana.
- “The farmer has to be successful, for us to be successful.” ~ Indra Kumar
- “He has been able to put up additional capacities at smaller costs than competitors due to in-house planning and execution,” ~ a senior manager who works closely with Kumar.
- The company follows project management techniques like Program Evaluation and Review Technique (PERT) and Critical Path Project Management (CPM).
- Indra Kumar plans for two to three years and then spends time executing the projects; in fact, he likes to keep an ear to the ground and often lives at the project sites.
- Avanti Feeds changed the way feed companies worked with farmers. Decided to do away with credit and switched to cash. “We have a simple theory. Credit kills. It tempts people and allows them to take their business casually,” ~ Indra Kumar. Farmers can now make better estimations of their yield and adjust their costs accordingly.
- “But from here onwards the company will take time to move to 10 lakh tonnes. The growth of the company is also a function of the capacity that it adds,” says an investor, who did not wish to be named but who has been invested in the company for the last 10 years. The general rule is that every ₹100 crore of investment in capacity expansion will result in ₹1,000 crore of additional sales.
- Is on the lookout for related areas to invest his funds. Currently, looking at new fish feed as well as new species to develop for the farmers.
- Considering cultivating sea bass, a protein-rich fish with a strong demand in the export market.
- Also looking to reduce the feed conversion ratio (the amount of feed given to the shrimps compared to their increase in weight). At present, 1.5 kg of feed generates 1 kg of shrimp; the company is trying to generate the same amount of shrimp with 1.3 kg of feed.
According to Indian reports, the European Commission’s visit to India starts today in Odisha. Officially the inspection by the Commission’s Health and Food Audits and Analysis team (formerly the Food and Veterinary Office, FVO) will be auditing the food safety official controls in place that relate specifically to fishery products. In view of the measures in place in the EU to protect consumers against antibiotic residues in farmed prawns, I think we can expect a certain amount of emphasis being placed in this area by the inspectors.
So what will the inspectors be looking at? Officially, the inspection will cover all aspects of food safety, and I think it will probably include a remit similar to that of the 2014 FVO audit:
“…to evaluate the performance of competent authorities and other officially authorised entities in their implementation of official controls concerning residues and contaminants in live animals and animal products, in order to assess whether these controls offer adequate assurance that the products and animals concerned, eligible for export to the European Union (EU) do not contain residues of veterinary medicinal products, pesticides and contaminants at concentrations in excess of EU maximum limits.”
The 2014 report is available here: http://ec.europa.eu/food/audits-analysis/audit_reports/details.cfm?rep_id=3325
The EU inspectors will concentrate in particular on the specific recommendations in the 2014 report. The relevant ones are:
Is there an adequate procedure that the Indian authorities use to carry out effective follow-up of non-compliant finds?
Are laboratories used for official controls adequate in terms of equipment, personnel, training, procedures certification etc.?
Have official labs improved their quality control and performance criteria since 2014, to ensure they are broadly equivalent to the performance criteria expected in European labs?
How good is awareness about antibiotic residues among relevant government officers, advisers and farmers?
Are official controls on the distribution and use of veterinary antibiotics effective? Are there procedures and controls that ensure that shrimps intended for the EU do not contain illegal residues?
Regarding the first recommendation on follow up, I would expect the European inspectors to ask about the outcomes of follow-up action taken to address the non-compliances fed back from the EU this year.
In addition there are some further clues in Decision 2016/1774, which is the piece of regulation that appeared in 2016 and introduced 50% testing of Indian shrimp at the European border. The concerns listed in the Decision refer back to the 2014 audit. There is reference to official controls on veterinary medicines being “very unsatisfactory”. The Decision says that:
“To date, the recommendations from the inspection report concerning official monitoring of aquaculture farms have not been satisfactorily addressed.”
And when will we hear the outcome of the visit? The European Commission understandably keeps all findings confidential until the report is published officially about 3 months down the line. However Indian sources may reveal some of the findings earlier, so it is worth keeping an eye on Indian press publications such as the Business Standard and The Hindu.
Source : An article by Ivan Barlow
‘According to Indian reports, the European Commission’s visit to India starts today in Odisha’
The 1st line is absolutely true. Avanti mentioned in its Q2 concall on 22nd that , 'they started from Paradip port ’
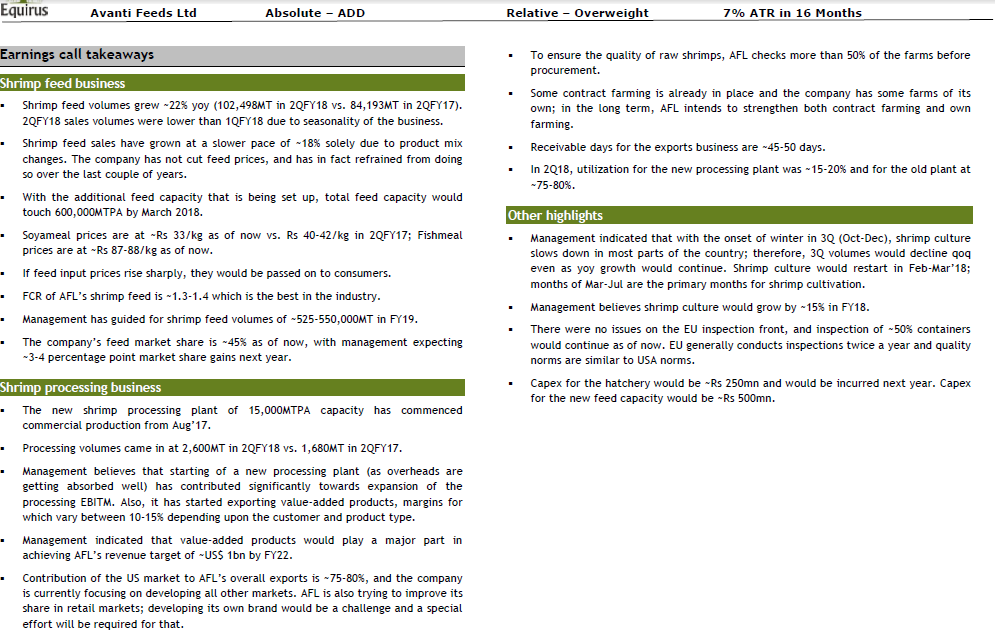
Equirus has come out with Q2FY18 Result Update for Avanti with Target price of 2950.
Avanti Feeds Q2fy18.pdf (724.9 KB)
Important piece of news in relation to European Inspection
AVANTI FEEDS LIMITED - Audio Conference Call conducted on 22nd November 2017
- Transcript of the Audio Conference Call
http://www.bseindia.com/xml-data/corpfiling/AttachLive/63f485a3-8966-4fbd-8f21-ac002c292db3.pdf
We may need to look at Chinese data also.
Foreign Trade Policy: Govt gives Rs 8,500 crore additional benefit to exporters
The incentive came as a part of the mid term review of Indias Foreign Trade Policy (FTP) 2015-2020.
US Shrimp import October '17 numbers are out… Stupendous volume growth continues…