The opening minutes of the conference call were a rehash of their investor presentation, found in the same document containing the results. The question and answer session gave some insight into the business. These are my notes, not a transcript, and I would check all figures against a transcript when released.
1) Colombia stock and sale should have started this quarter, and in Mexico/Brasil should start soon. Can you tell us how much revenue you’re expecting from these…
Starting a warehouse in these countries. Waiting for the covid situation to get better. Looking to put up the warehouse in the next 3-6 months. Want to visit these countries to do our surveys as soon as we can.
2) Expectation for FY22 from the US:
Target is to achieve Rs. 125-135 Cr.
3) Why did you choose to put the oncology injectable plant in a different subsidiary?
We’re putting the plant as a subsidiary for income tax purposes.
A certain Mr. Kacholia came on to ask the next few questions.
4) Regulatory compliances - What steps have you taken to remain on top of the compliance curve?
- We hire external consultants. We work with 2 former FDA inspectors that have had over 35 year stints with the FDA. They conduct trainings on data integrity, quality assurance and conduct mock inspections.
- We try to take people from quality compliant companies, especially when we hire people in the semi regulated markets.
- Going into much higher levels of automation, with the use of limbs for example, and going completely paperless. We’re investing in software such as eBMR, and those will go some ways to ensure compliance will be high.
5) What is the total potential output from the plant, what is the utilisation in order to reach 135Cr.
We’re at around 40-45% capacity utilisation, but since we’re expecting approvals for many products, we want to make sure there’s no bottleneck for new approvals. Therefore we’re doubling our current capacity in the next twelve months
6) How much is this capacity expansion, will you invest it all at once?
Will cost us 110Cr. This is the entire phase II of our injectables. Since Caplin is sitting on a large reserve, we’re going to complete the CapEx with this, and we’re not getting external investment from companies like Fidelity.
7) Is the equity structure going to remain the same? Is it 70/30?
It is 75/25 and will remain exactly the same. The extra capital will be in the form of debt from parent to subsidiary.
8) How much is the total new capex + work in progress?
300Cr in terms of gross block.
9) What is the total revenue potential at full capacity?
Target is revnue of $100mil(732 Cr.) from this plant by 2026. We will start looking for high quality contract manufacturing as well.
10) Is there any opportunity for us to play in contract vaccine manufacturing?
Vaccines come under the category of biological products, we are in pharmaceuticals. Due to regulatory rules, we can’t produce one in a plant that’s meant for the other. It would take time to set this up. We evaluated this, but decided not to proceed. We also hear there’s significant capacity for vaccine injectables coming in around the Hyderabad region in the next twelve months.
11) Do you expect growth in LatAm to continue?
Within LatAm, we’re in the smaller geographies, moving to larger geographies like Mexico. We are not focusing on products like anti HIV. Instead, we are going to work on 25 APIs, each giving a smaller piece. This gives us the opportunity to undercut local suppliers while not competing with bigger players. If bigger players enter this space, we’ll undercut their costs.
We’ve also noticed another trend. When latin American people travel to the US, they carry with them supplies for their families. We’ve noticed they like our products and carry our generics. Our goal in 5-6 years is that we also build a brand among the LatAm people living in the US and sell a range of products to them.
12) Debtor days and inventories? Explain. What’s the difference in the debtor days in US.
In the US, is between 45-60 days. Current receivables are around 80-100 depending on the time of the year. As US is starting to become a larger portion of our business, you’ll see the number coming down.
On inventory days, there are three kinds of inventory. Inventory in the factory is not a good sign. You have inventory in the waters, and inventory near the market. Inventory in the market, especially during covid times is a boon. Raw materials are not coming to local suppliers there. We are the only ones with warehouses there. Plus, we don’t have 1-2 products but 300-500. Our products have sold there for 15 years. I would prefer the inventory to be on the higher side.
13) What about the USA?
Today, 100% of our sales in the US comes from our partners. We don’t keep any inventories in the US and the model there is quite conventional. We want to change this by 2023, introducing a frontend.
14) As we scale up the complexity in the business, what have you done to hedge yourself against the currency fluctuations in the countries you’re operating in:
Panama, El Salvador and Ecuador, the currency is dollar. Guatemala is the country which gives us the most business. The currency there is stable. Nicaragua, Honduras and others have not had issues with volatility. The one country that isn’t stable is the Dominican Republic. If our model was based on giving credit to customers, we’d be at risk. However, since our products are in the warehouse, we can adjust the prices for fluctuations and sell the products at variable prices to the customer to hedge.
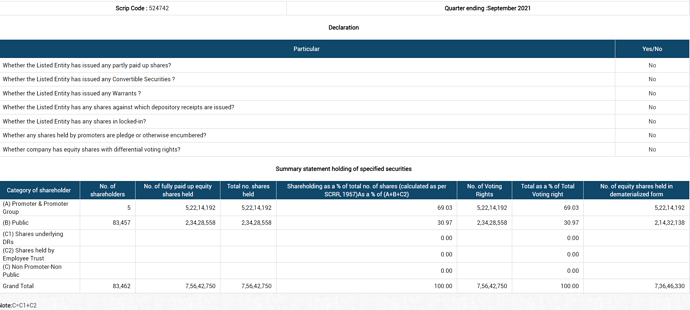
15) Can you explain Caplin Steriles to us. What’s the revenue breakup between licensing and sale. What was the EBITDA and margins for FY21?
Export revenues 58%. PAT was -22Cr. EBITDA is -24 Cr.
16) You’re planning CapEx for Caplin Steriles, and sending money from the parent company’s reserves, but looking at the annual report from 2020, there is 150Cr lying in the balance sheet, and around 125Cr lying around this year. Why do we need more money from the parent company?
The investment from Fidelity is enough to run the business. However for CapEx, we take this money in from the parent company. We learnt a lesson in the last 18 months, US is approving things quickly, and spare cash is needed for approvals. Don’t want to be left behind, so we’re keeping that money. If there was to be no CapEx, all OpEx is taken care of.
17) What’s the target for Caplin Steriles in a few years?
Filing charges and R&D is pulling down the numbers. Without those, the numbers are similar to the parent company. Once we develop the DMF and start the front end in 2023, we’ll be completely vertically integrated and the bottom line will reflect this.
18) Oncology plant status?
We acquired buildings, so the process should be done a lot quicker and we don’t need construction. We expect the first batch of oral solids in 12-15 months. On API, it’ll take 2 years for a greenfield project. If it is inorganic, it’ll be much quicker, but we’re working on the assumption that it’s organic right now. So the 300-350Cr. will be spent over the next 24 months.
So they seem to be giving a lot of importance to their warehouse model, stocking up on inventory close to the customers. Secondly, they’re keen on opening their own front end store in the US by 2023, and that’s also the timeline for their backward integration to make some progress. They’ve given quite a target for their revenue from the US, as they want to go from 130Cr. to roughly 750Cr. in five years.
I’m posting this not as a bull thesis, but to keep a record on VP forums to compare future results and statements. Personally, I have the following questions in the LatAm regions:
- Lets work on the premise that the management can’t be trusted. Are there any studies of field testing pharmaceuticals in the LatAm regions that can give us an understanding of the quality of their products? I found https://www.usp.org/global-public-health/medicines-quality-database which does the following in Africa, LatAm and the Carribean:
The majority of the medicines in MQDB are screened initially in the field by PQM-trained personnel following standardized procedures. Medicines undergo physical and visual inspection initially and subsequently analytical tests, currently consisting of disintegration and thin layer chromatography.
Update: a comprehensive search across the database returned two hits. One particular batch/lot in 2011 failed quality inspections, given below. The tests concluded that the medicines are not counterfeit. There have been no failed checks since. The questions now are to understand the details of when a drug fails a compendial test, and what conclusions one can draw from this. They haven’t received an OAI or such a warning from the FDA. I’m more than happy to write to the company regarding this.
- Does anyone know more about the competitive and regulatory landscape in the various LatAm countries? I don’t want to make generic statements given that the story should be completely different across borders. I found a study by Flood et al. (2017), which looks at the perception and utilisation of generics in Guatemala. I’ll post an update later when I’ve understood more.