Govt halts plan:
Several US hospitals teaming up to manufacture their own generics and sell at no profit. Doesn’t help the case of Indian generic pharma companies.
The most significant manufacturing costs in generic manufacturing are api and labor. The US has to be dependent on Low cost countries like India in both these aspects. Such a move in my opinion will only help Indian companies as more business will be outsourced to us. Given the distributor consolidation that is happening/ has happened in US which actually brought down margins of generic manufacturers one more customer to sell to will only help Indian Pharma companies as to that extent customer bargaining power comes down.
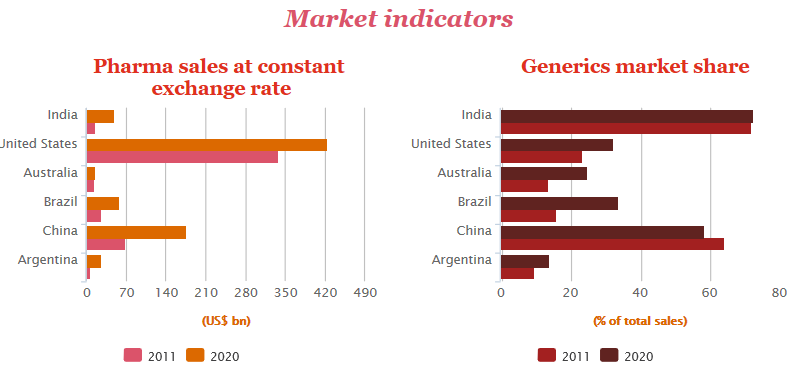
Top Bullish points on Indian pharmaceuticals
• India’s cost of production is nearly 33 per cent lower than that of the US
• Labour costs are 50–55 per cent cheaper than in Western countries. The cost of setting up a production plant in India is 40 per cent lower than in Western countries
• Cost-efficiency continues to create opportunities for Indian companies in emerging markets & Africa
• India has a skilled workforce as well as high managerial & technical competence in comparison to its peers in Asia
• India has the 2nd largest number of USFDA-approved manufacturing plants outside the US
• India has 2,633 FDA-approved drug products. India has over 546 USFDA-approved company sites, the highest number outside the US
Source: India brand equity foundation
Bearish POINTS
More USFDA approvals do not mean better margins for Indian ‘big pharma’
Indian pharma is in a transformative phase following the pricing pressure on generic drugs in its key market, the US. While most firms are making a transition to less competitive complex generics and speciality drugs
Source: Cadila health Annual report
On can find an Excellent article : → India's leadership in cost-efficient generics is unchallenged: Sanjiv Kaul | Business Standard News
Regards
Sector analysis HDFC 2018Q4
@ananth I could not locate any separate thread for Sun Pharma. Is there a separate thread or kindly point to the best person who can provide an outlook on Sun Pharma. I am invested heavily in the stock but the stock price has been depressed for a considerable length of time. I have not yet averaged because I feel that the stock has not yet bottomed out (technically speaking) …
Will appreciate any insight into the company and its latest result. The price has taken a beating today.
Though they claim it as for Pharma industries, does this covers only the institute’s recommended stocks ? For me it seems so, at least majority of the list are generic pharma companies. No mention of some prominent players like Ajanta, Natco, Biocon, GSK etc.
More difficulties for pharma companies in US.
dear shenbal,
i am also in similar predicament. Only having bought the stock yesterday at 480. My close relatives worked with rambaxy for several years and currrently in pharma space only The clear view is that for any new R&D work the existing prices of drugs have to be higher. If the governments continue suppressing prices our future generations will have to suffer. Sun pharma has not bottomed out. I think the bottom is always farther than we think. I thought it to be 500 but I thnk it would be nearer to 400. I have been observing it for close to a year,but it seem to have taken support around 500.
anyway 2018,19,20 will be tougher for both the company and investors. If we take this as an oppurtunity missed @7% per year then its ok. otherwise you got to stay away from this sector and this stock.
Seems like many of Trumps’ announcements this may be more smoke than fire. Proposal is focussed more on reducing middle men than directly negotiating down prices or price control with drug makers.
When will we ever have stable rules in this country.
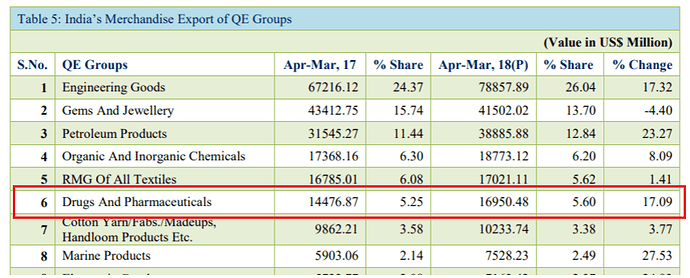
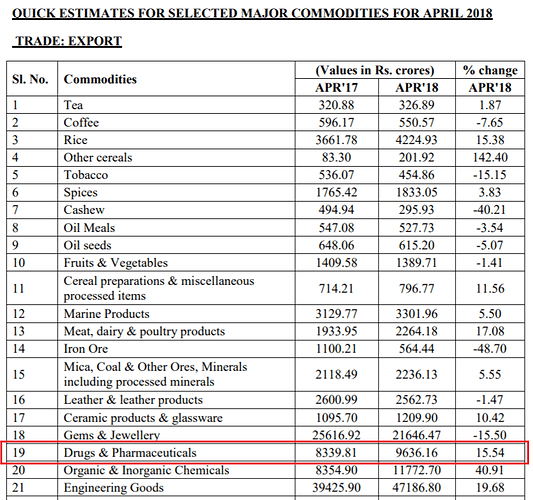
Is pharma export data telling us something?
Pharma export from India up by 17% in Fy18 over Fy17 (in USD value terms).
Source: http://dgftcom.nic.in/exim/2000/tradestat/mis/bapr18.pdf
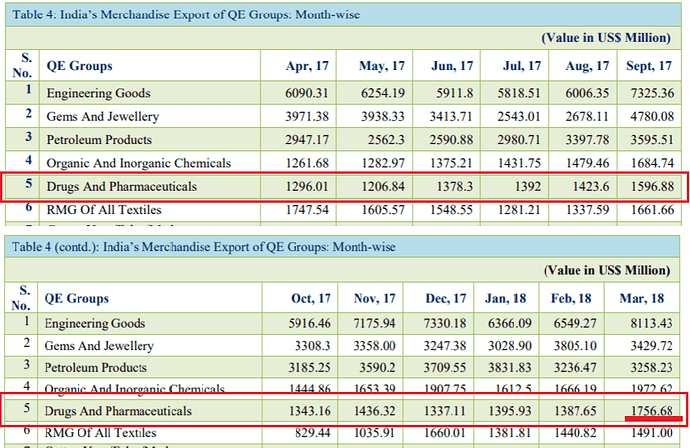
Monthly trend. Good spike seen in Mar '18 numbers.
Followed by YoY 15.54% jump in April 2018 (per estimates data; in INR value terms)
Thanks Sandeep , today saw 28% growth in export data n I was wondering same. Has Pharma n Textile started contributing back. This is very helpful
Trade War: India to raise duty on 30 US goods
This seems like a very bad idea.
Investing in software and pharma might not be a great idea now. Never know if and when they will be hit with tariffs.
I am speculating by the way, so please take it with a pinch of salt.
Buyer-consolidation and heightened-competition have been the primary reasons for the rapid price erosion in US generic base business for pharma cos.
What caused heightened competition? GDUFA (introduced in 2013-14). It increased pace of ANDA approvals.
Prior GDUFA (i.e. 2007-14), ANDA approval rate was constant at ~500 per year, resulting in golden years for generic pharma cos.
Note: FDA’s fiscal year (FY) runs from Oct 1 to Sep 30. *FY2018 data is for 8 months from Oct '17 to May '18.
Seems ANDA filing rush has been moderating. Can see ANDA Receipt/Filing (TTM) rate becoming constant or start to dip a bit. Additionally, ANDA withdrawal rate has been rising - new trend. Together, these two offers possibility of stagnant new ANDA approval rate (say ~750 per year) going forward. If it happens, then it could soothe price erosion in base business in times to come.
Raw data source: FDA site
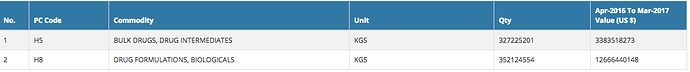
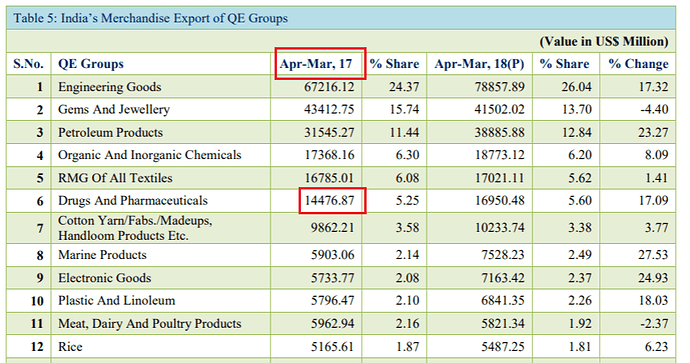
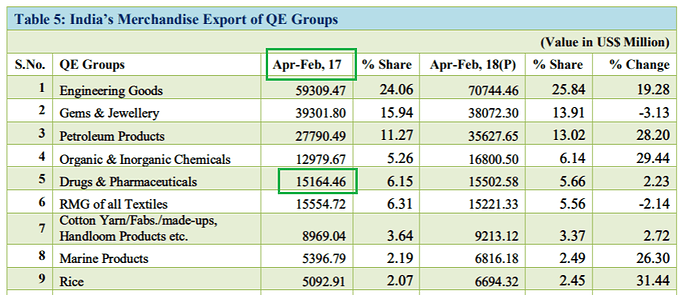
I am a bit confused by these numbers. I think the Drugs and Pharmaceuticals group includes ‘Bulk Drugs and Drug Intermediates’ and ‘Drugs Formulations, Biologicals’ in the export data as the sum of the numbers matches quite closely.
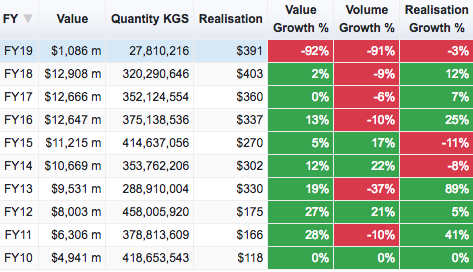
Bulk Drugs
Formulations
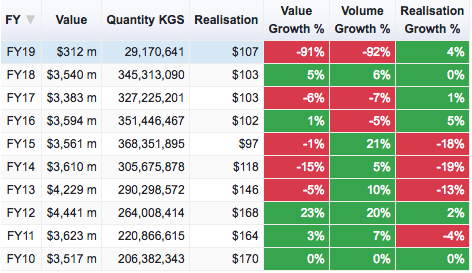
If you see FY17 over FY18, neither of these have shown a 17% uptick in exports. Both together would be around 3-4% at best. FY17 numbers of these two heads stand at USD 16,049 million (well over the 14476.87 reported in that table).
Not sure which of these numbers are wrong. My numbers are from
http://121.241.212.143/principalcommditysearch.html
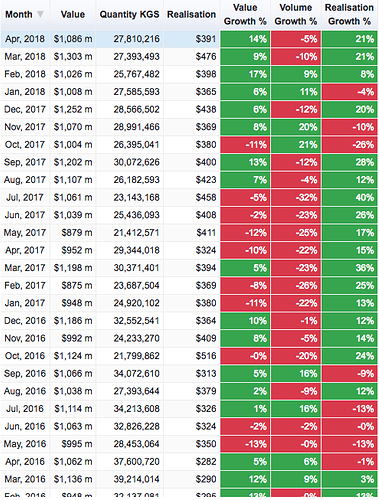
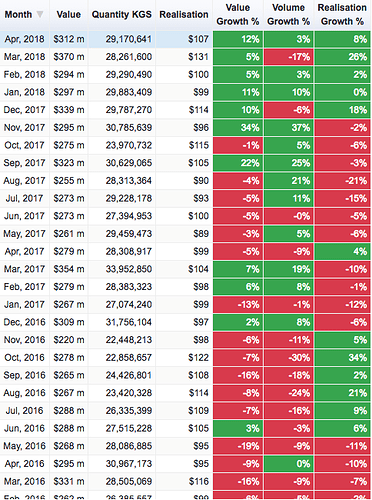
As for monthly numbers, this is how it looks for these two heads (bulk drugs and formulations).
There is a bit of an uptick in the last 6 months but its just a 8-10% growth in Q4 over a low base so I think there isn’t much to get excited about. But again, one of these data sources is wrong or my assumption (that Bulk Drugs and Formulations make up the numbers under Drugs & Pharmaceuticals in the report) is incorrect.
Thanks @phreakv6, for illustrating data from an alternate source.
Found the error source; the following snapshot has incorrect base data for Apr-Mar’17 (marked in red).
Basically, Apr-Mar’17 number (source here) cannot be lower than Apr-Feb’17 number (source here). Lower base has resulted in incorrect hard to believe 17% growth number.
Having said that, your post illustrate interesting numbers for last 6 months in both categories i.e. bulk drugs and formulations.
healthpolicy.usc.edu/documents/USC%20Schaeffer_Flow%20of%20Money_2017.pdf
well documented piece on how the US Distribution system works
Now, coming to the point on generics, yes the patent cliff of 2009-2014 was like the Y2K moent for Indian Pharma companies as they took full advantage of the first wave of generic penetration. Now with those low hanging fruits out of the way, generic manufacturers have a limited market opportunity to play in
Would this shift focus to the API piece as Volatility of raw materials means that simple blending companies who would source their RM from china and just blend them to make the final API start witnessing disruptions in their business?